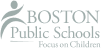Ozone Bleaching
Ozone is a toxic gas generated by applying a high voltage to oxygen. The first commercial use of ozone for bleaching was in 1992, so ozone is relatively new for pulp bleaching. However, ozone provides several economic and environmental benefits over other chemical options for elemental chlorine free (ECF) and totally chlorine free (TCF) bleaching. This course discusses the benefits of ozone bleaching, its chemistry, how it is generated, and the process and equipment involved in ozone bleaching.








Demos + Pricing
Learn more about our courses, get pricing, and see our platform.
Course Details
Learning Objectives
• State the benefits of ozone bleaching (compared to other chemistries)
• Describe ozone bleaching chemistry
• Explain how ozone is generated
• Explain how ozone bleaches pulp
• Identify important ozone bleaching variables
• Identify and describe ozone bleaching processes and equipment
• Identify and describe safety hazards and safe work practices associated with ozone bleaching
Specs
Frequently Asked Questions
How is ozone generated?
What are the advantages of ozone bleaching?
Does ozone bleaching require special equipment?
What process variables are important for ozone bleaching?
Is ozone hazardous?
Sample Video Transcript
Because ozone gas is unstable and cannot be stored, it is manufactured-on-demand. Ozone is created by applying high voltage to a gas that contains oxygen. For pulp bleaching, pure oxygen is used so a high concentration, 10 to 12% of ozone can be generated. Electrons travel across a gap between electrodes. When oxygen molecules collide with the electrons, they break apart into individual oxygen atoms. The individual atoms attach to molecular oxygen and form ozone.